Chitinozoans are extinct microfossils widely recorded in the Ordovician to Devonian strata (ca. 480~359 Ma) and playing important roles in biostratigraphy. However, their biological affinity is still debated since the first description in 1931.
Generally, previous hypotheses about their biological affinity fall into two main camps since 1930s: first of all, relationship with various groups of protists, which mostly consist of ideas based on the vesicle morphology during the 1930s to 1980s. However, none of these hypotheses are totally convincing because of the hermetically sealed vesicle, which seems to disable any communication between the inner chamber of chitinozoans and the environment, and thus precludes the possibility of obtaining food. Secondly, eggs of unknown metazoans, a hypothesis generated by the discovery of the cocoon-like preservation during the 1960s to 1980s. The "egg hypothesis" was subsequently reinforced by the perception that there was no fossil record of immature individuals or evidence of any reproductive process in chitinozoan assemblages. A recent study has, however, questioned the prevailing "egg hypothesis" by documenting a large morphological variation in chitinozoan populations that is inconsistent with the variation found in modern and fossil eggs of aquatic invertebrates, and thus suggested that chitinozoans were independent microorganisms (Liang et al., 2019).
Recently, 20 exceptionally preserved specimens were discovered or re-examined among hundreds of thousands of specimens processed from the Middle and Late Ordovician (ca. 467~445 m.y. old) limestones of Laurentia and Baltica. Associate researcher LIANG Yan from the Nanjing Institute of Geology and Palaeontology, Chinese Academy of Sciences (NIGPAS), and other paleontologists from Estonia, the United States, the United Kingdom and Central South University, have carried out new research on the biological properties of chitinozoans and gained new understanding. This study has been published online of Geology.
The inner structures of these unusual specimens were analysed by a novel combination of imaging techniques, including the near-infrared microscopy (NIR), the focused ion beam scanning electron microscopy (FIB-SEM) and X-ray micro-computed tomography (micro-CT). These unusual specimens (Figs. 1, 2) are turned out to precisely record successive reproductive moments—the missing reproduction stages in the life history of chitinozoans.
Studies of the inner ultrastructure (Figs. 3, 4) are carried out and spongy materials have been detected for the first time. Together with the test wall, those spongy materials are turned out to be made of clustered rounded spherical particles with a diameter around several tens to over one hundred nanometers, which are the basic material building the organic test of chitinozoans.
The reproductive strategies—hitherto undescribed, which produce either one or several offspring at a time, have been discussed and reconstructed (Fig. 5). The reproduction resembles budding and strobilation, representatively, but with significant differences.
The morphology, geometric shapes, significant within-population variation, widespread distribution in disparate paleoenvironments, and the new evidence for two modes of asexual reproduction make it plausible to link chitinozoans, at least the taxa presented in this study, with protists.
Financial support was provided by the National Natural Science Foundation of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, the Estonian Research Council, and the State Key Laboratory of Palaeobiology and Stratigraphy, Nanjing Institute of Geology and Palaeontology.
Reference:
Liang, Y.*, Hints, O., Tang, P., Cai, C.Y., Goldman, D., Nolvak, J., Tihelka, E., Pang, K., Bernardo, J., Wang, W.H.*, 2020. Fossilized reproductive modes reveal a protistan affinity of Chitinozoa. Geology, v. 48. https://doi.org/10.1130/G47865.1.
Liang, Y.*, Bernardo, J., Goldman, D., Nolvak, J., Tang, P., Wang, W., Hints, O.*, 2019. Morphological variation suggests that chitinozoans may be fossils of individual microorganisms rather than metazoan eggs. Proceedings of the Royal Society B 286, http://dx.doi.org/10.1098/rspb.2019.1270.
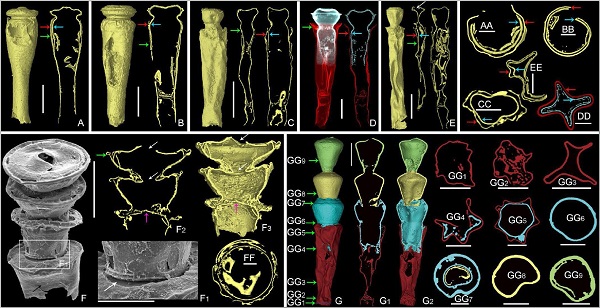
Fig. 1 Reproductive specimens under Micro-CT. The offspring test can be obviously observed in the chamber of the parental specimen

Fig. 2 NIR (A, L) and FIB-SEM analyses of the reproducing chitinozoans
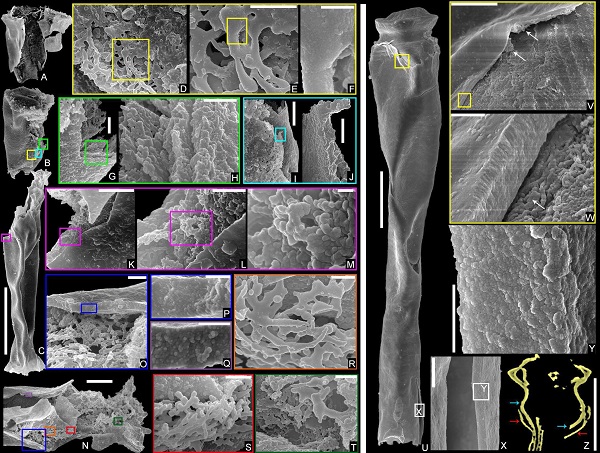
Fig. 3 Morphological ultrastructure of reproductive specimens

Fig. 4 Ultrastructure of the sponge structure (left) and the test wall (right)
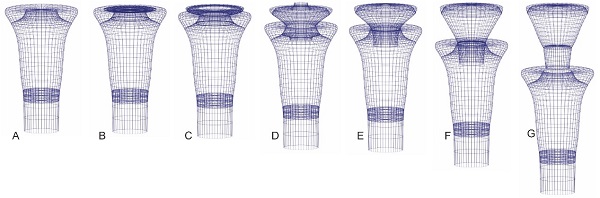
Fig. 5 Hypothetical 3D perspective drawing of the reproductive cycle of Cyathochitina campanulaeformis (Fig. 2L)
